Question 5#
A 24-year-old male was admitted to the surgical ICU following open reduction and internal fixation of a tibial fracture after symptoms consistent with a transient ischemic attack in phase II of recovery. All workup thus far has remained inconclusive of a cause to include MRI/MRA, echocardiogram, telemetry, and EEG. The next day he has downtrending hemoglobin and platelets, an increase in serum creatinine, and a temperature of 38.8 without leukocytosis or leukopenia. His surgical site is without any obvious sign of infection. Physical examination is remarkable for petechiae seen all over his body. Blood smear reveals schistocytes. ADAMTS13 levels were noted to be <10%.
What is the most appropriate treatment for this condition?
A. Discontinue all antibioticsB. Stop all heparin products and start argatroban
C. Plasma exchange
D. IVIG and high-dose corticosteroids
Correct Answer is C
Comment:
Correct Answer: C
Antibiotic-related side effects have not been reported to cause the myriad of symptoms presented in this case. IVIG and high-dose corticosteroids are not used for the management of TTP but can be used for various other immunologic reactions including idiopathic thrombocytopenic purpura (ITP). ITP is autoimmune destruction of platelets without activation of platelets, would cause bleeding, would result in petechiae/purpura but would not cause symptoms of organ ischemia as in this patient, schistocytes, or decreased vWF. Argatroban would be appropriate if the patient had HIT. The patient’s 4T score is not consistent with HIT. His symptoms are most consistent with a thrombotic microangiopathic anemia.
Thrombotic Microangiopathic Anemias (TMAs) present with a classic triad or pentad of symptoms. The triad consists of fever, anemia, and thrombocytopenia, whereas the pentad includes renal and neurologic dysfunction. The three common etiologies of TMA are Hemolytic Uremic Syndrome (HUS), atypical Hemolytic Uremic Syndrome (aHUS), and Thrombocytopenic Thrombotic Purpura (TTP). There are but a few findings that can reliably differentiate among them table below:
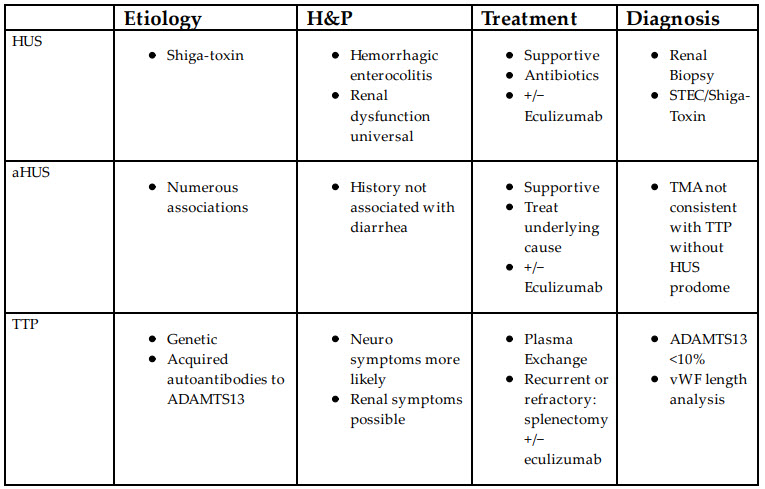
TTP is a result of abnormal, absent, or autoimmune disruption of ADAMTS13 (vWF-cleaving protease). Antibodies to ADAMTS13 are responsible for >90% of the diagnosed cases of TTP. Known causes of autoimmune TTP include ticlopidine and HIV. TTP is also associated with malignancies and various infectious processes. Most cases of acquired ADAMTS13 deficiency are idiopathic.
The pathophysiology of TTP is a result of dysfunction of ADAMTS13, resulting in dysfunctional vWF. vWF is excreted from Weibel-Palade as a long-coiled chain. ADAMTS13 cleaves this chain into smaller molecules. As a result of ADAMTS13 deficiency, longer vWF more readily provokes platelet aggregation. Ultimately, the inability of ADAMTS13 to properly process vWF results in abnormal clot formation in microvasculature and subsequent organ ischemia.
Consistent with its pathophysiology, confirmation relies upon ADAMTS13 levels <10% and analysis of vWF showing a greater proportion of long vWF multimers compared to controls. However, these tests are unusual and may have an unacceptably long processing time. Relying on history, symptoms and rule-out of other disease processes is crucial for timely treatment. Furthermore, improvement in symptoms will be seen with plasma exchange in TTP, will not affect HUS outcomes, and may help with aHUS. There may also be a role for eculizumab in the treatment of TTP.
Untreated, TTP is mostly fatal in 10 to 14 days. Plasma exchange is the treatment of choice. The exchange provides ADAMTS13 and reduces circulating autoantibodies. Giving plasma can be a temporizing measure. Various immunosuppressive regimens have been reported, though data do not support their use at this time. Splenectomy is a last resort for those who are unresponsive to plasma exchange or are reliant on plasma exchange due to repeated occurrence; data on outcomes are sparse.
References:
- Kaur H, Sasapu A, Fox MH, Motwani P. Successful eculizumab therapy in thrombotic thrombocytopenic purpura (TTP) refractory to plasma exchange, steroids and rituximab. Blood. 2014;124(21):2794. Available at http://www.bloodjournal.org/content/124/21/2794?sso-checked=true.
- George JN. How I treat patients with thrombotic thrombocytopenic purpura: 2010. Blood. 2010;116(20):4060-4069. Available at http://www.ncbi.nlm.nih.gov/pubmed/10942361.
- Tsai H-M. Thrombotic thrombocytopenic purpura: a thrombotic disorder caused by ADAMTS13 deficiency. Hematol Oncol Clin North Am. 2007;21(4):609-632. Available at https://www.sciencedirect.com/science/article/pii/S0889858807000706? via%3Dihub.
- Dubois L, Gray DK. Case series: splenectomy: does it still play a role in the management of thrombotic thrombocytopenic purpura? Can J Surg. 2010;53(5):349-355. Available at http://www.ncbi.nlm.nih.gov/pubmed/20858382.
- Schwartz J, Eldor A, Szold A. Laparoscopic splenectomy in patients with refractory or relapsing thrombotic thrombocytopenic purpura. Arch Surg. 2001;136(11):1236. Available at http://archsurg.jamanetwork.com/article.aspx? doi=10.1001/archsurg.136.11.1236.